Medicines Health & Regulatory Authority this week sent letters to UK CBD businesses informing them they must stop supplying CBD products by November 1st until their products have received Marketing Authorisation.
In the mean time this will result in the suffering of thousands of patients with hundreds of medical conditions being unable to access their medicine.
Here is what the MHRA had to say about the matter officially.
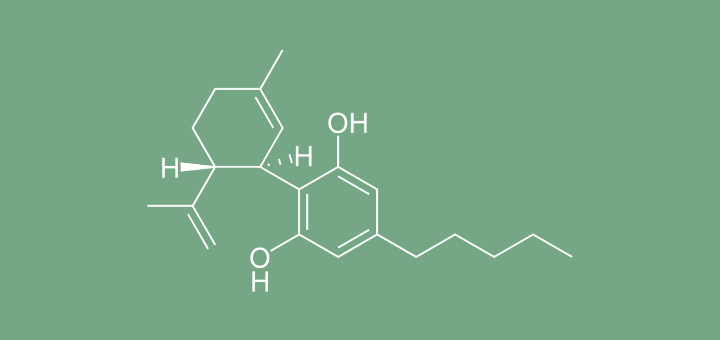
“We have come to the opinion that products containing cannabidiol (CBD) are a medicine. Products for therapeutic use must have a medicines’ licence before they can be legally sold or supplied in the UK. Products will have to meet safety, quality and effectiveness standards to protect public health.
“If you use CBD and if you have any questions, speak to your GP or other healthcare professional.
“We have written to UK CBD stockists and manufacturers to inform them of our view. These products will require a marketing authorisation to be granted before they can be legally sold, supplied or anywhere advertised in the UK.
“We can provide regulatory guidance to any company who may wish to apply for a licence.”
To read the letter sent to CBD businesses in the UK and to understand more about why CBD is being considered a medicine in the UK please check our previous article.
What do we do?
From here we must take direct action. We have set up a petition with 38 Degrees to try and raise a wider awareness of the rash decision that MHRA are making which will directly negatively impact some of the most vulnerable patients in society. Patients with seizure disorders, those treating inflammatory diseases, as well as PTSD, diabetes and MS are already wondering how they are going to access their chosen CBD products.
Sign our petition:
Stop MHRA Removing CBD Products From Sale! Patients Will Suffer!
Please share the petition on your social media pages!
Share this from our Facebook and Like us.


Comments (3)