How do we work around the problem of adhering to some of the most strict medical guidelines in the world for medicine and at the same time help those that need it without causing any more harm?
In recent years the use of medicinal cannabis has become a subject of much debate with global political powers now looking to re-evaluate their stance on the plant and its molecules. Within the UK, calls for medicalisation of cannabis provoked the British government to legislate for access to medical cannabis for UK patients from the 1st of November 2018.
Despite this new legislation, it remains exceedingly difficult for UK patients to access medical cannabis products through the NHS. The governing body that provides national guidance and advice to NHS professionals is the National Institute for Health and Care Excellence (NICE).

NICE provides a series of evidence-based guidelines that outline the best practices for UK medical professionals based on the extensive medical evidence and studies that are available to a committee of clinical experts. Medical professionals in the UK must adhere extremely closely to the NICE guidelines as they protect them from a legal standpoint when navigating throughout all aspects of medicine. These guidelines are developed based on evidence built up over years of surgical procedures, clinical trials and experimentation. Given this, cannabis poses a particularly sticky situation for all involved.
For decades medical research into cannabis was discontinued following a sustained campaign from the American Medical Association in 1945 that forbade exploration of cannabis for medical use. This later resulted in the classification of cannabis as a substance of abuse leaving cannabis research in its infancy for many years leaving a wealth of unexplored potential. Though there has been a recent renewal of medical cannabis research, compared to opiates and opioids, we are some thirty to forty years behind in our understanding of cannabis medicines. This is the key issue currently facing NICE and the NHS.
Although the government has technically opened the door to medical cannabis, NICE currently lacks the double-blind placebo-controlled drug testing required to develop medical guidelines for medical cannabis. This has generated a standstill in the accessibility of medical cannabis, ultimately stemming from the historic ban on research. This has meant we are decades behind in our understanding of this exciting branch of reawakened medicine. The current blame game surrounding medical cannabis has generated a fog of misinformation and chaos for patients across the UK. Despite the lack of costly double-blind drug trials there still exists a wealth of published medical data demonstrating the vast potential of cannabis as a therapy in various diseases.
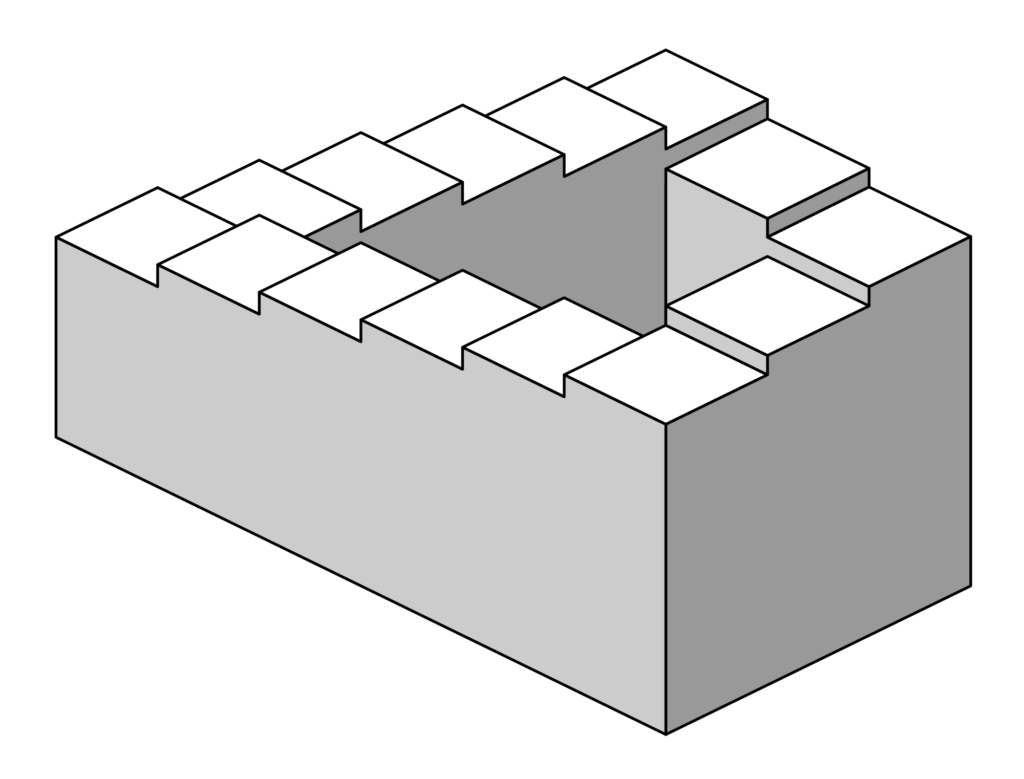
The aim of this article is to highlight this issue and provide support to those currently caught within this loop of frustration that resembles the Penrose steps. The first rational step then would be to initiate research programmes that aim to add clarity to the situation and answer the unknowns that currently inhibit NICE guidance and public access. This raises another issue. To develop a body of evidence this substantial it would require decades of clinical trials, and billions of pounds in research funding would need to be invested in cannabis research. All the while patients around the UK would still be unable to access the potential of these treatments.
How do we resolve this?
Cannabis constituents are less toxic to humans than water and are extremely well tolerated by the body. Questions raised about the safety, and efficacy of cannabidiol have been quelled by studies showing prolonged exposure to cannabinoids both in animals and humans noting the absence of adverse effects (Fine and Rosenfeld, 2013).


This debate boils down to several key medical principles of ethics and human rights: respect for autonomy, beneficence, non-maleficence and justice. The current medical cannabis legislature directly infringes and inhibits the fulfilment of these core medical principles. Again, this is not the fault of medical professionals, as they have been placed in this difficult and potential career-risking scenario in which they are being asked to provide patients with informed consent without having guidance themselves. The current reluctance to prescribe cannabis based the principle of “do no harm” may in fact be having the opposite of the desired effect. This ability to withhold treatments due to potential risks compromises the autonomy of the patient who may derive significant benefit from a cannabis treatment.
Fundamentally this choice of treatment and course of management resides with the patient or guardian and not with the medical professionals or legislators. This fundamental right is currently being infringed and in light of the significant evidence supporting medical cannabis it is unethical to withhold such treatments if there is grounds for its experimental use. Perhaps the most impacting step would be to remove this burden from medical professionals through regulated public access. This public access would mean that research professionals can conduct observational research while giving patients the opportunity to trial cannabis based medicines alongside current treatment regimes. Based purely on just how safe and well tolerated cannabis is compared to current legal prescription and recreational drugs, it is more than reasonable to consider the potential of implementing a strictly regulated medical cannabis system (Sachs, McGlade and Yurgelun-Todd, 2015); a system that addresses and carefully monitors and minimises many of the risks we already know are associated with crude cannabis use.
MUST READ: A List Of All The Studies On Cannabis And Epilepsy (Over 100!)
This article is an extract from our upcoming handbook which has been compiled to ensure patients are armed with the information they need to understand just exactly what benefits medical cannabis may or may not provide them. This article does not promote cannabis as a treatment for all illnesses but instead aims to highlight that there is more to medical cannabis than just the cannabis sativa plant. Cannabis has shone a light on a world of exciting medical potential in the form of endocannabinoids and the endocannabinoid system which has untold therapeutic potential across all mammalian health and disease.